题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
[主观题]
Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g),已知在298K时,Ag2O(s)的 =-30.6kJ·mol
Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g),已知在298K时,Ag2O(s)的 =-30.6kJ·mol
Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g),已知在298K时,Ag2O(s)的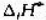 =-30.6kJ·mol-1,
=-30.6kJ·mol-1,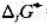 =-11.2kJ·mol-1,试求在298K时
=-11.2kJ·mol-1,试求在298K时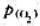 的压力是多少(Pa)?Ag2O(s)的热分解温度是多少?(105Pa下)
的压力是多少(Pa)?Ag2O(s)的热分解温度是多少?(105Pa下)
查看答案
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案


 更多“Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g)…”相关的问题
更多“Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g)…”相关的问题
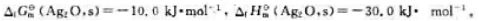
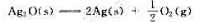
 的
的 、
、 的
的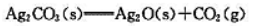
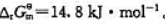 383K烘干时,空气中加入多少CO2可避免Ag2CO3分解?
383K烘干时,空气中加入多少CO2可避免Ag2CO3分解?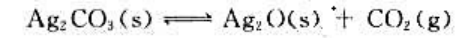
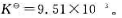 现将Ag2CO3(s)放入烘箱中,在383.15K下干燥,问空气中CO2的体积分数最小为何值时才能避免Ag2CO3(s)分解?
现将Ag2CO3(s)放入烘箱中,在383.15K下干燥,问空气中CO2的体积分数最小为何值时才能避免Ag2CO3(s)分解?