题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
1000K时,反应C(s)+2H2(g)==CH4(g)的△rGmΘ=19 290J/mol。现有与碳反应的气体,其中含有CH4(g)10%,H2
1000K时,反应C(s)+2H2(g)==CH4(g)的△rGmΘ=19 290J/mol。现有与碳反应的气体,其中含有CH4(g)10%,H2(g)80%,N2(g)10%(体积%)。试问: (1)T=1000K,P=100kPa时,甲烷能否形成? (2)在(1)的条件下,压力需增加到多少,上述合成甲烷的反应才可能进行?
 答案
答案
×
(1)△G=△rGmΘ+RTlnQp=19290+8.314×1000×ln0.156=3843.4J△G>0甲烷不能形成。(2)若合成甲烷的反应能进行,则须KpΘ>Qp△rGmΘ=一RnnKpΘ19290=一8.314×1000×lnKpΘKpΘ=0.09825P>159.03kPa
 如果结果不匹配,请 联系老师 获取答案
如果结果不匹配,请 联系老师 获取答案

 更多“1000K时,反应C(s)+2H2(g)==CH4(g)的△…”相关的问题
更多“1000K时,反应C(s)+2H2(g)==CH4(g)的△…”相关的问题
 CH4(g)的A,Gf=19290/mol.现有与碳反应的气体,其中含有CH.(g)10%,H2(g)80%,N2(g)10%(体积%)。试问:
CH4(g)的A,Gf=19290/mol.现有与碳反应的气体,其中含有CH.(g)10%,H2(g)80%,N2(g)10%(体积%)。试问: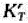 =1.00,若于密闭反应罐中通入1×105Pa的水汽与足量的红热碳反应,试确定平衡时诸气体的压力和水汽的转化率.
=1.00,若于密闭反应罐中通入1×105Pa的水汽与足量的红热碳反应,试确定平衡时诸气体的压力和水汽的转化率.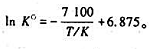 已知原料中只有反应物A(g)和过盘的B(s)。
已知原料中只有反应物A(g)和过盘的B(s)。
 的Kp=4×10-2,则反应HBr(g)
的Kp=4×10-2,则反应HBr(g) 1/2H2(g)+1/2 Br2(g)的Kp是( )。
1/2H2(g)+1/2 Br2(g)的Kp是( )。 2SO3(g)在1000K时的KƟ=3.4×10-5,计算1100K时的KƟ。已知该反应的ΔrHƟm=-189kJ/mol,并设在此温度范围内ΔrHƟm只为常数。
2SO3(g)在1000K时的KƟ=3.4×10-5,计算1100K时的KƟ。已知该反应的ΔrHƟm=-189kJ/mol,并设在此温度范围内ΔrHƟm只为常数。
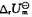 和标准摩尔反应热力学能
和标准摩尔反应热力学能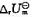 。
。